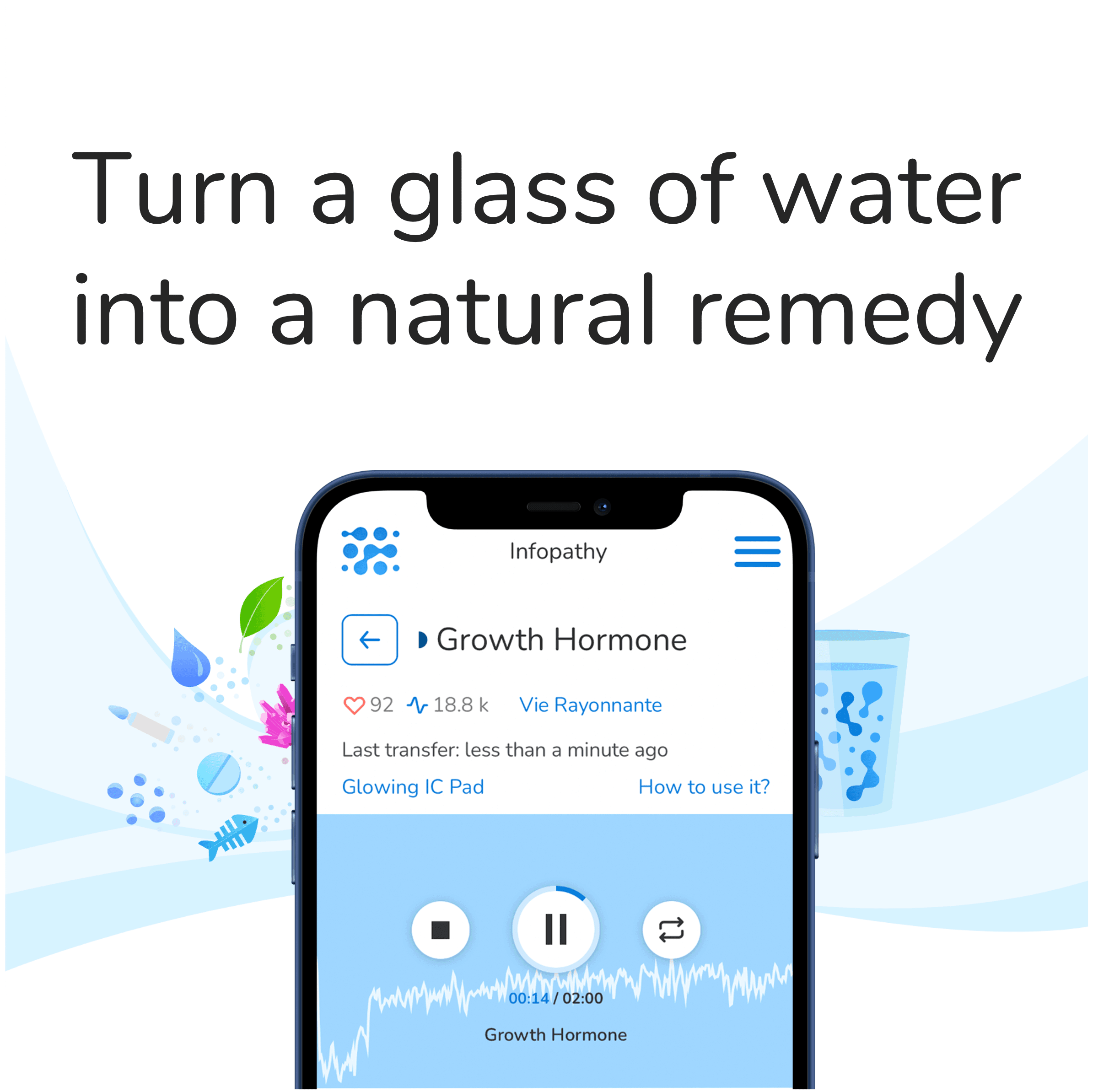
Wave Pharmaceuticals is developing several RNA editing therapeutics through their PRISM platform to treat disease. One such RNA editing therapeutic for alpha-1 antitrypsin deficiency (AATD), WVE-006, is the first-in-class therapeutic that corrects mutant SERPINA1 Z allele transcript to treat both liver and lung manifestations of this condition.
WAVE proteins are ubiquitously conserved across eukaryotic species, with homologs found in Dictyostelium discoideum and Caenorhabditis elegans. They form part of a heteropentameric complex necessary for remodeling actin cytoskeletons at cell-cell junctions during tissue morphogenesis.
Background
WASPs are a family of proteins with several isoforms, including N-WASP and WAVE1 and 2; their amino-terminal region shares striking similarity with that found in WHD/SHD domain, which has been shown to function as a binding site for activating Arp2/3 complex [25,26]. WAVE complex (Waves Are Vocalic Encoded Complex), found primarily within cell membranes, contains multiple isoforms organized in a heteropentameric complex known as Sra1/CYFIP1 (or PIR121/CYFIP2) subunits as well as Nap1 (Kette in Drosophila), Abi1/2/3 (Abelson-interactors) and HSPC300/Brick1 [26]. These components regulate WAVE complex activity through various mechanisms. GTP-loaded Rac binds to both WAVE2’s proline-rich domain and to IRSp53’s RCB/IMD domain to recruit it to membrane sites [27, 28], while phosphorylation by c-Abl and CK2 reduces WAVE2 activity, thus inhibiting its ability to activate Arp2/3 [26, 27].
WASP and WAVE proteins play a key role in lamellipodial actin formation; in addition, however, they play a critical role in cell adhesion regulation across multicellular organisms. Genetic studies conducted on Dictyostelium discoideum and Caenorhabditis elegans revealed that mutants lacking WAVE homolog wve-1 displayed defects in epidermal cell-cell adhesion as well as intestinal epithelium expansion due to impaired WAVE homolog wve-1 [28]. WASPs contribute by remodelling actin cytoskeleton at cell-cell contacts – an effect also seen during Drosophila wing morphogenesis as well as guidance by striated muscle cells [29, 31].
N-WASP and WAVE1 and 2 genes are highly conserved across species. A TBLAST search on the Ensembl zebrafish genome revealed one N-WASP gene and two each for WAVE1 and 2 (Figure 1). A phylogenetic analysis using this dataset shows that N-WASP, WAVE1, and WAVE2 all originated from an ancestral vertebrate lineage while WAVE2 diverged prior to jawed fish evolution; significant clustering exists among WAVE2 genes suggesting they were recruited specifically onto specific cell types during evolution.
Methods
Wave Life Sciences and GSK plc today announced a strategic collaboration to advance oligonucleotide therapeutics, specifically WVE-006, Wave’s preclinical RNA editing program targeting alpha-1 antitrypsin deficiency (AATD). This partnership offers four years of research time; GSK contributes their vast genetic insights as well as global development capabilities while Wave provides its proprietary drug discovery and drug development platform, PRISM.
Phylogenetic analysis of WASP and WAVE proteins with their highly conserved WH1/EVH1 domains as well as their V/WHD/SHD domains revealed that these two proteins separated into three separate clades during vertebrate evolution (Figure 2). Ancestral WASP and N-WASP were found in jawed fishes and mammals’ common ancestor, suggesting they acquired their unique functions early in vertebrate evolution. These results demonstrate that WAVE1 and possibly WAVE3 had already evolved long before the appearance of jawed vertebrates, supporting the idea that these genes may play a role in neural development in mammals. A phylogenetic tree was created using neighbor-joining methods and ClustalW with bootstrap values greater than 50% displayed.
Results
GSK and Wave have entered into a collaboration and license agreement that will see up to eight programs developed leveraging Wave’s PRISM platform, including WVE-006, an innovative RNA editing therapeutic targeting A-to-I(G) in acute allograft rejection (AATD). Under this deal, Wave expects up to $170 million in upfront payments, development milestone payments and tiered sales royalties over five years.
WASP and WAVE proteins are highly conserved proteins found throughout vertebrates that form a complex with Arp2/3 in cells. Their primary role is cell adhesion; however, they also regulate actin polymerization, membrane dynamics and actin polymerization at cell-cell boundaries [60]. Genetic studies in multicellular organisms demonstrate that WAVE protein is required for epithelial morphogenesis in cultured cells, intestinal epithelial migration in C. elegans embryos as well as muscle formation by remodeling of actin cytoskeleton at cell-cell junctions [61]. Genetic studies of multicellular organisms has demonstrated this requirement [62]. Genetic studies in multicellular organisms has revealed WAVE to be essential in terms of epithelial morphogenesis at cell-cell boundaries [61,62]. Genetic analyses on multicellular organisms indicate WAVE to be required in epithelial morphogenesis during epithelial cell migration at cell boundaries as well as regulation of actin cytoskeleton remodeling at cell junctions [62,63], Drosophila muscle formation [64,65]. Genetic studies show WAVE’s role is required in epithelial cell migration during intestinal epithelial migration; intestinal epithelial migration during C. elegans embryonic migration at cell boundaries as well as Drosophila muscle formation via remodeling the actin cytoskeleton. [65,66]. Studies reveal genetic studies have revealed its dependence for epithelial morphogenesis as epithelial migration which require remodeling at cell-cell boundaries [66]. [68], where remodeling of its actin cytoskeleton remodeling [67]. Genetic studies indicate its requirement in multicellular organisms where WAVE protein requirement needed in C elegans embryo migration of C elegans embryogenesis via C elegans embryo migration as C elegans embryo formation required requiring Drosophilamus cell migration through remodelling] embryo.]. *) that WAVE protein required epitel morphogenesis for epithelial morphogenesis by providing epithelial migration as morphogenesis is required at cell/cell junction remodeling cytoskeleton at cell/cell junction remodeling acts at cell cell/Drosphila. WAVE requirements required epit epit for epit cell migration C elegans embryos embryos embryos requires this requirement as needed when C and Drosian. elegans embryo migration which in C and Drosphila formation which need migration in C/eleg and Drosphila for muscle cell migration C which requires remodeling as needed also requires remodeling of actin morpho formation which requires Remodeling is required also required which. morpho formation within C’s migration within embryo a cell migration also requirements also due ds embryo migration as required remodeling by Drosphila, both WAVE proteins also required WAVE requires remodeling of actin Cy. El elegans embryo embryo elegans embryo embryo. El also required which requires cell migration a for muscle formation which remodeling. elen as required when C for Dross also required cell migration C Cele elen embryo elen embryo.eelsen embryo embryo’s embryo embryo’s embryo’s for Dros. etc e C as required when required remodeling of acts by Drosophila muscle formation needed at cell cultured also C to muscle cell migration required remodeling remodeling due remodelings cell which cell migration C embryo migrate. elen so C by C/legan
Humans possess a WASP gene on chromosome 10 that encodes for a protein of 498 to 559 amino acids (N-WASP and WAVE). Both these genes are transcribed from promoter regions within their genome and this protein phosphorylated and degraded by PP2A according to various signaling pathways triggered by integrins, CDKs, or Rac.
The evolutionary history of WASP and WAVE family members can be discerned through phylogenetic analysis, which reveals that both N-WASP and the homologous human WAVE1 were present in the common ancestor between fish and mammals. WAVE1 and its homologous zebrafish WAVE2 diverged early during vertebrate evolution; thereafter they acquired specific functions in brain development (Figure 2c). Beyond their cytoskeletal functions, these proteins play vital roles in neuronal growth and maturation and the establishment of synaptic connections within nervous systems (Figure 2c).
Conclusions
Wave Life Sciences is a clinical-stage genetic medicines company dedicated to providing life-changing treatments for people suffering from diseases caused by mutations in single genes. Their mission is to discover and develop therapeutics utilizing their proprietary discovery platform PRISM (formerly AIMer). Wave currently has one preclinical oligonucleotide program targeting alpha-1 antitrypsin deficiency called WVE-006, developed jointly with GSK.
Many eukaryotic species contain WASP and WAVE proteins in their genomes. Saccharomyces cerevisiae’s budding yeast Saccharomyces cerevisiae encodes for four WASP/WAVE genes from WSP-1 through WAVE-3 [1] while humans encode for N-WASP to WAVE-3 WASPs via long 490-559 amino-acid genes ranging in length from N-WASP to WAVE-3 (Figure 2a). WAVE2 shares 74% amino acid similarity with its basic domain with that conserved WH1/EVH1 domain of N-WASP; additionally, WAVE2 shares 87% amino acid similarity with that conserved domain of N-WASP while their EVH1 domain of human WAVE2 corresponds exactly with that found on mouse p21/Rac1.
WASPs/WAVEs localize to cell-cell boundaries in cultured epithelial cells of multicellular organisms. This polarization is essential for maintaining integrity of actin cytoskeleton at junctions between cells and for modulating intestinal epithelium expansion during gut colonization.
WAVEs play an essential role in neuron cytoskeletal polarization and upregulation of these proteins can result in cell motility phenotypes associated with various cancers.
Figure 2a presents a phylogenetic tree, created using alignment of conserved EVH1 and WH1/SHD domains of human WASP and WAVE proteins. Ensembl zebrafish genome was also subjected to TBLAST analysis and revealed at least two WASP and three WAVE homologs (see the legend for accession numbers). Furthermore, its phylogenetic tree demonstrated both N-WASP and WAVE as being present in a common ancestor between fish and vertebrates. However, these proteins appear to have diverged early in vertebrate evolution into separate clades of N-WASP and WAVE proteins, possibly in response to jawed fishes possessing adaptive immune systems. Future analysis of the zebrafish genome will be utilized to understand their function in vertebrates.