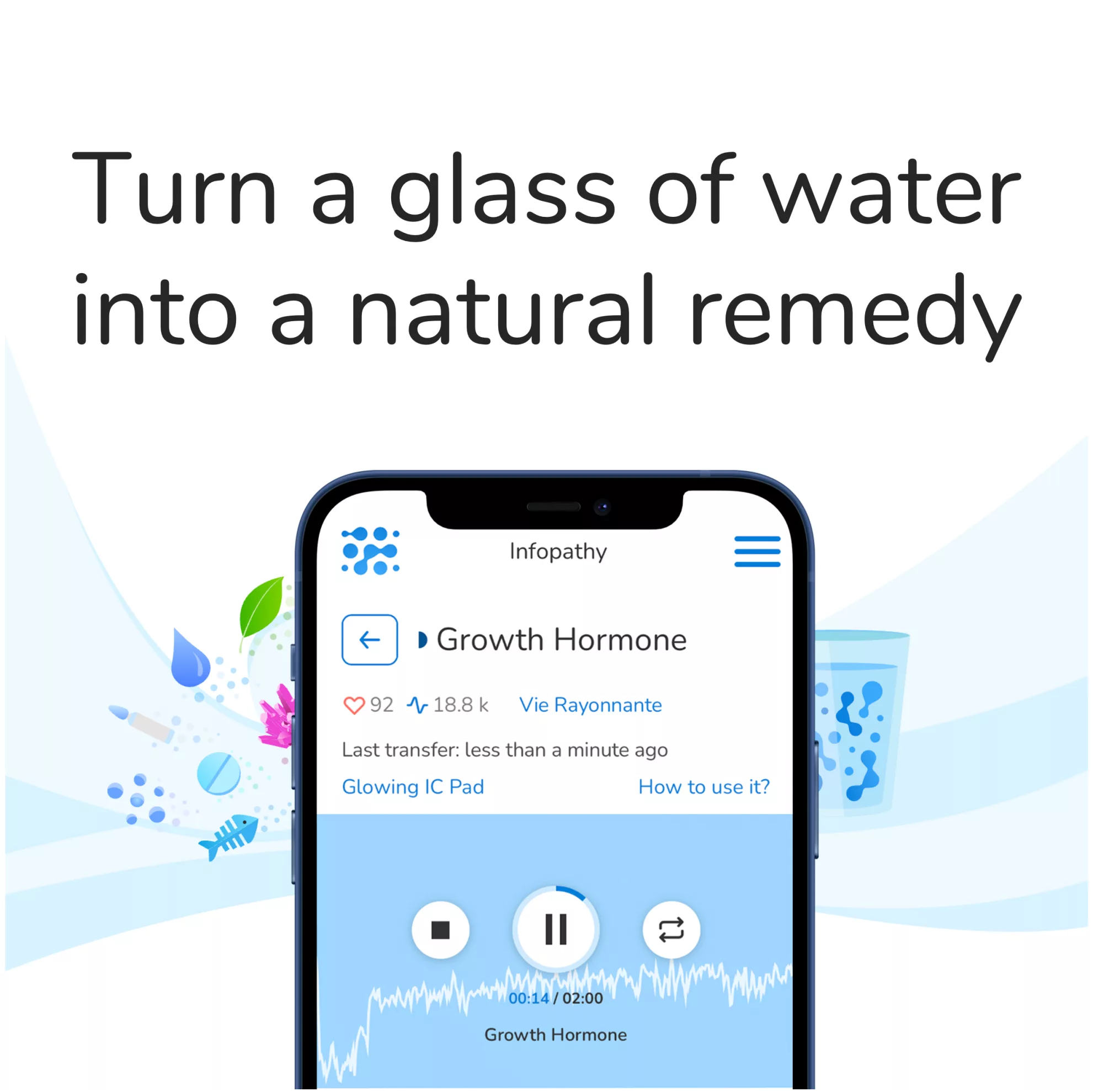
Wave Genome Ra is an innovative new approach for identifying pathways associated with RA using gene expression data. It uses GWAS, gene expression analysis and adaptive permutation tests to find those significantly associated with the condition.
Core promoter architecture is distinguished by an abundance of TATA boxes7 and sharp transcription initiation profiles.8, in contrast with miR-430 genes which exhibit more open chromatin signals at 256 cell stage.
What is a wave genome?
The Wave Genome Hypothesis proposes that waves of gene expression appear, peak, and fade sequentially during early development. Each wave corresponds to a genomic sequence or chromosome which contains multiple genes with unique activation patterns – ZGA (Zygotic Gene Activation) is usually observed during 2-cell to 4-cell embryo stages while minor waves peak between sphere/dome stage and morula stage; all waves are initiated by single enhancers.
MiR-430 cluster and other members of the minor wave become active during ZGA wave activation due to open chromatin states and histone modification marks like H3K27ac. Publicly available epigenomic datasets representing ATAC-seq chromatin opening, transcriptional activity states in early development (RNA-seq), and presence of cis-regulatory elements (ChIP-seq) were utilized to define DNA sequence features and promoter architectures that distinguish minor wave genes from their neighbors.
MiR-430 genes proximal to transcription bodies exhibit a distinct promoter architecture in which no sharp transcription start site (TSS) exists and instead a broad promoter region extends inward towards transcription start site. These promoter regions may be targeted by H3K27ac targeting histone modifications, and possess unique core promoter architecture from those activated during the zygotic phase.
Histone modification mark H3K27ac was significantly less prevalent in miR-430 promoter regions compared with CG sets, suggesting that their unique activation profiles might be related to its lower abundance in these promoter regions; suggesting a more restrictive environment is required for their expression.
WAVE GENOME LLC provides one of the world’s most advanced rejuvenation treatments: A Digital Biohologram of each client’s Wave Matrix is coded onto a microchip and read by a bio-holographic TESLA generator, transmitting nanolevel signals through nanoholography to client cells to promote profound increases in health and rejuvenation of entire organism. TESLA GENOME is the result of years of research by some of the world’s most advanced scientific groups including Bauman Russian State Technological University (MIT level), former USSR research institutes specialising in psychotronics research institutes.
What is a wave genome ra?
Wave Genome LLC has designed a new generation of Psi-Generators that are based on electret technology, used for decades in Russia with great success. However, unlike previous devices that simply coded electrets with frequency drugs or supplements for rejuvenation purposes, Wave Genome LLC pioneers bioholographic wave treatments for rejuvenation purposes using an electret device containing each client’s wave matrix encoded as biohologram and delivered as part of this treatment protocol.
These personalized digital drugs are tailored specifically for each client according to Dr Irene Caesar’s theory of Wave Optics in Chromosomes, sold either through Tesla Human Cell Digital Pharmacy (TESLA HUMAN CELL DIGITAL PHARMACY); or Remote Laser Treatment, depending on their choice.
What is a wave genome pg?
WASP and WAVE proteins play an integral role in various actin-based cell structures, including endocytic vesicles, filopodia and podosomes/invadopodia. How these diverse architectural shapes are constructed through similar molecular actions is still one of the great mysteries in cell biology; their discovery may hold important ramifications for tumor growth, cell motility and blood vessel formation during embryonic development.
Recently, it has become clear that N-WASP plays an essential role in the formation of different structures by recruiting other proteins to form complex networks that sculpt actin cytoskeleton. N-WASP forms a homopentameric protein complex with Sra1 and Nap1, two other members of WAVE family; Abelson interactors Abi1/2/3 (also known as Nap2 in Drosophila); its core is made up of proline-rich VCA domain which simultaneously binds two proteins at once to initiate polymerization initiation of actin polymerization initiation of actin polymerization of actin polymerization initiation.
N-WASP and WAVE proteins also interact with GTPase Rho, which facilitates reorganization of actin cytoskeleton in an intricate fashion influenced by additional signaling pathways. Rac can directly bind to WAVE1 and WAVE2 basic domains via its SH3 domain to recruit them to membrane. GTP-loaded Rac may also bind with the RCB/IMD domain of IRSp53 from the EF-C/F-BAR family and thus indirectly regulate WAVEs through this interaction.
Though the origins of N-WASP and its family remain obscure, phylogenetic analyses based on alignment of highly conserved WH1/EVH1 domains indicate they originated prior to divergence of eukaryotic cells. Additionally, identification of WAVE and WASP homologs in plants suggests that development of Arp2/3-based actin polymerizing machinery was essential in multicellularity evolution.
What is a wave genome cg?
Wave genome Cg (WGCg) is an embryonic stem cell gene whose promoter is activated by pioneering transcription factors, including Nanog in Drosophila8, Pou5f3 and Sox19b in Xenopus9,8,10, BRD4 in human13 have all been demonstrated as key core promoter elements during ZGA13,15 binding H3K27ac marks on chromatin then activating major waves 16-17 of ZGA based on TATA box presence and sharp transcription start site (TSS) profiles16-17
This evidence supports the theory that major and minor waves are two distinct genomic activation events and that their emergence is managed by specific promoter architectures. Therefore, it should come as no surprise that major wave genes tend to respond better to pioneering factor binding than minor wave genes.
Additionally, we observed that minor wave genes were highly enriched for nascent RNA and active RNA Polymerase II speckles while major wave genes showed minimal enrichment; this suggests that their respective chromatin environments were more conducive to transcription than that around major wave genes; consistent with previous findings that permissive transcription states are correlated with reduced genome mutation rates.
At our lab, the differences between major and minor wave genes were further investigated using sequence analyses of promoter architecture. Major wave genes displayed broad TSS profiles while minor wave genes displayed more limited ones with canonical TATA boxes and canonical TSS profiles. Furthermore, we observed that only minor wave genes managed to avoid global transcriptional repression while major wave ones could not.
Minor wave genes often exhibit more limited promoter regions and lack TATA-box-dependent, sharp TSS core promoter codes than major wave genes; as a result, minor wave genes may not respond directly to pioneering factors through interactions between TATA-box dependent promoter sequences and pioneering factors; on the other hand, major wave genes tend to respond better in such instances.